Bone Therapeutics is moving forward with a new phase II trial targeting severe osteroporosis – but this time with its allogeneic bone cell therapy (ALLOB). This type of therapy is easier to administrate, and could reach more patients in need.
 Bone Therapeutics is based in Gosselies (Belgium) and is developing cell therapies to treat conditions like bone fractures, osteocronosis and severe osteoporosis.
Bone Therapeutics is based in Gosselies (Belgium) and is developing cell therapies to treat conditions like bone fractures, osteocronosis and severe osteoporosis.
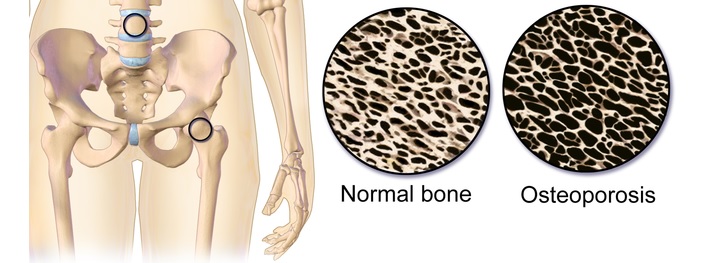
Of these indications, osteoporosis is a main focus. This disease is considered a serious public health concern – and an ageing population will continue to increase the number of people affected.
The total market estimated to be worth up to €7.4Bn ($8.4Bn), of which up to 30% is deemed severe osteoporosis – that does not respond to available therapies. Bone Therapeutics’ cell therapy would particularly help this subset of patients.
The company is now transitioning to allogeneic development in osteroporosis, by focusing on a new phase II trial with ALLOB – their off-the-shelf product. The preparation of the trial is already in place, and recruitment should start in the beginning of 2017.
This decision follows the promising efficacy results for its autologous therapy (PREOB) in a phase IIa trial for osteoporosis. Additionally, ALLOB has recently shown a good safety profile in a trial for a different indication.
The CEO of Bone Therapeutics (Enrico Bastianelli) has said that ALLOB is a better solution for patients that its autologous counterpart. It seems that PREOB will not go forward to a Phase III in osteoporosis…
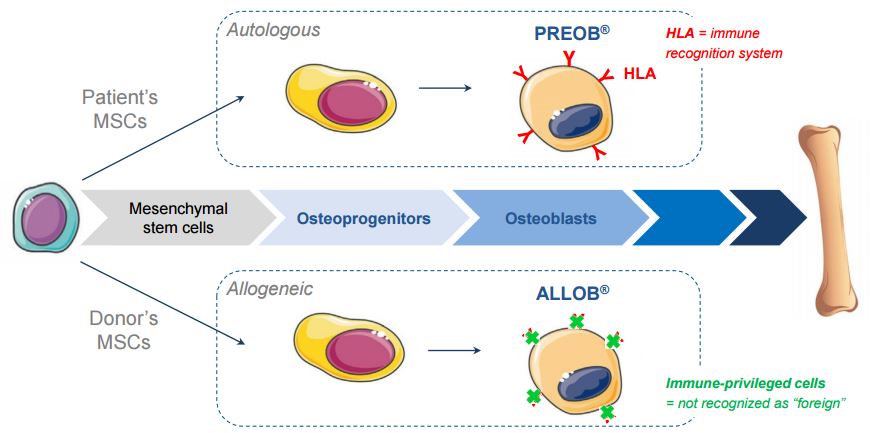
The move towards allogeneic makes sense, as the therapy can be used right away by patients in need.
In industrial terms, an allogeneic therapy also has more potential to be scaled up, as opposed to a personalized procedure in autologous therapies. In this way, it can reach much more patients – a point that the CEO of Cellectis, the universal CAR-T developer, highlights in this interview.
With this new trial, ALLOB is now being studied in 4 conditions. Could this shift to ALLOB in a big indication (osteoporosis) mean that Bone Therapeutics is on its way to become the Biotech for universal bone cell therapy?
Featured Image Credit: Cross section of hip bone (CC 2.0 Patrick Siemer)





