CAR-T therapies have gained plenty of attention lately by promising to end cancer with a one-off treatment. But all that glitters is not gold; experts remind us it is important to be critical about the immuno-oncology hype.
Immuno-oncology has become all the rage lately, and the hype has led to some major investments, acquisitions, deals and partnerships. It seems that the label “immuno-oncology” is sufficient to increase the value of the product. But Dr. Philip Greenberg, cofounder of Juno Therapeutics, a big Biotech developing CAR-T therapies, has called attention to the many challenges this field faces.
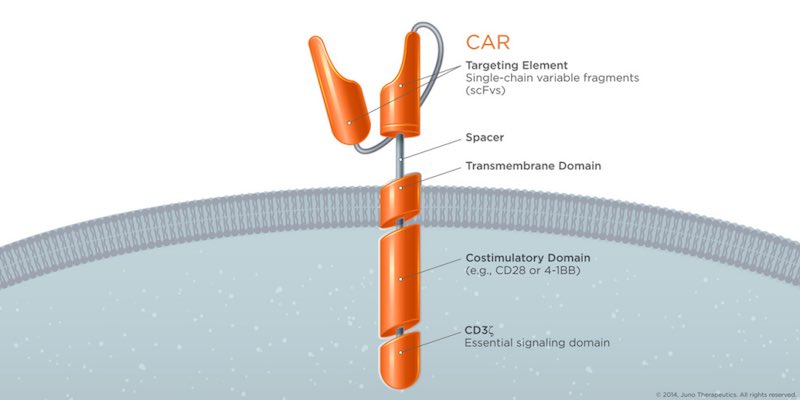
The first attempts to stimulate T-cells to attack tumors got the attention of big Pharma like Novartis, Kite Pharma and Juno Therapeutics. These companies are now advancing their programs in CAR-T, the breakthrough therapy that everyone’s after. In this treatment, the patient’s T-cells are engineered in vitro, rewired to attack a specific tumor antigen and then reintroduced in the patient (for more details, check out our review!).
But despite the promising initial results, most patients with cancer have shown relapses and no long-term improvement. In addition, the treatment can have very severe side-effects that not all patients will be able to withstand in their weakened state.
One of them is the cytokine release syndrome; when T-cells attack a tumor, they secrete such high amounts of inflammatory cytokines that the whole body reacts with intense fever, nausea, fatigue and swelling.
Another major complication is neurotoxicity, which manifests as hallucinations, memory loss and brain swelling. These are in fact the symptoms that presented the three patients that died in Juno’s Phase II trials earlier this year. But in spite of the current risks of the technology, Novartis and Kite Pharma are racing to be the first to gain FDA approval for their CAR-T therapies, with Juno at their heels.
The difficulty of treating tumors mainly relies on their ability to rapidly mutate and acquire drug-resistance, hide from the immune system, suppress the immune response and even fight back. And they don’t make an exception with CAR-T. In some cases, after a first attack from the engineered T-cells, tumors are able to eliminate the target antigen.
The huge patient variability is also a major issue that is not yet properly addressed. Recently, a Nature article highlighted how successful outcomes in precision oncology are usually over-represented in the media against the thousands of cases where the technology hasn’t proven effective.
We don’t mean to be pessimistic, but rather realistic. By being down to earth and discerning which approaches work from which don’t, scientists will be able to accelerate the development of successful immuno-oncology strategies.
On the bright side, some treatments have been successful in treating a broad range of patients; in Germany, a CAR-T study recently showed remission rates of up to 94% in blood cancers. And many other companies are already working in circumventing the current limitations of immuno-oncology so that they can reach more patients.

For example, researchers from the Dutch ISA Pharmaceuticals recently highlighted that successful immunotherapies specifically target the immunosuppressive mechanisms of tumors. This can be achieved by combining the treatment with chemotherapy or checkpoint blocking.
Celyad, in Belgium, is developing a unique CAR-T therapy directed to a target that is involved in the modulation of the tumor microenvironment and present in 80% of cancer types (for more info, check our interview with Georges Rawadi, its VP Business Development).
Stimunity, in Paris, focuses in the 70-80% of patients that are under the risk of CAR-T’s severe side effects due to their weak immune systems. To address the problem, this company targets an immune response with a lower inflammatory profile.
The French Cellectis became famous last year when its CAR-T therapy saved a 1-year old girl from leukemia. Cellectis’ approach to make CAR-T safer is based in the implementation of a switch control system; CAR-T cells are inactive by default and only get activated when rapamycin is added. In June this year, the first patient was treated in Phase I trials with its candidate drug UCART19.
Finally, although CAR-T therapies are currently dominating the immuno-oncology field, some companies are exploring alternatives. That is the case of Vaximm, which is developing a treatment that uses bacteria to deliver tumor-specific antigens.

Our conclusion is pretty much summed up in Cellectis’ CEO words:
I’m just trying to be realistic, CAR-T is not the miracle cure for cancer.
Meaning that there is still a lot of work to be done until this therapy can be safe and effective for the majority of patients. In addition, CAR-T therapies also face a big challenge in terms of marketing and sales.
A source we talked to yesterday mentioned that the estimated price of these treatments rounds the €0.5M-1M range. This will make it difficult for companies to get reimbursements, though the high incidence of cancer may account for that. The results from Kite Pharma and Novartis will soon set a benchmark for other companies if they do manage to launch their products early next year.
Featured image credit: Lightspring/shutterstock.com
Figure 1 credit: Juno Therapeutics
Figure 2 credit: Sciencepics/shutterstock.com
Figure 3 credit: Robert Kneschke/shutterstock.com