Ipsen is paying Exelixis $200M upfront, plus $545M in future milestones to acquire commercialization rights for a multi-organ cancer candidate outside US, Canada and Japan.
California-based Exelixis has found an European partner to license the small molecule drug, cabozantinib. Ipsen, a French biotech group, will have comercialization rights for cabozantinib worldwide, except in the US and Canada (where Exelixis retains its operation) and Japan (where it has another partner).
This deal cost Ipsen about €184M ($200M) upfront.
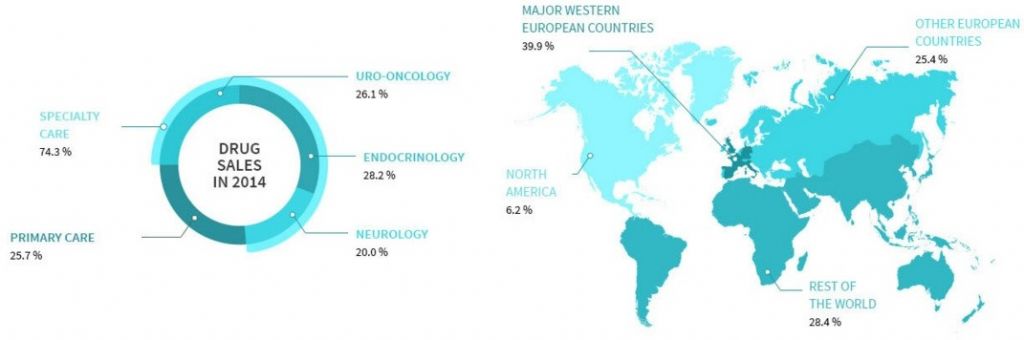
Ipsen mainly specializes in neurotoxins as therapies (for diseases that cause spasms and neurological conditions). However, it has also recently been moving more into the oncology field.
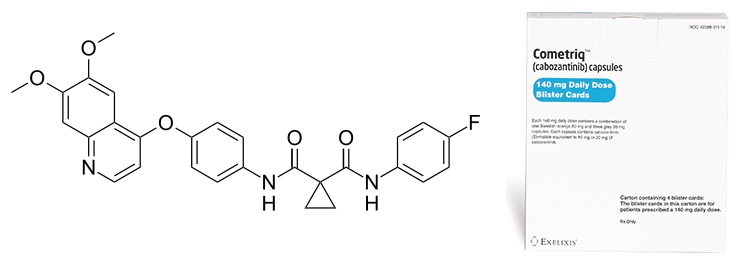
Cabozantinib is already approved as a therapy for metastatic medullary thyroid cancer (MTC), under the commercial name COMETRIQ. However, this drug does have the potential to become a global cancer treatment…
Cabozantinib is in advanced investigation for other cancers, which form the basis of the future milestone payments – and these can reach up to about €500M ($545M).
It has also finished a phase III trial for renal cell carcinoma (RCC), which obtained good results and was published in the New England Journal of Medicine. So, the use of cabozantinib for RCC is waiting for approval in the US and EU.
Exelixis is also conducting a phase III trial for hepatocellular carcinoma (HCC), which is currently one of the deadliest cancers and for which there is still a therapy void.
Besides the market for this 3 cancers, the two companies are looking forward to explore more targets.
Exelixis reports that they have more than 45 ongoing or planned studies – in areas like advanced renal cell, bladder, colorectal, non-small cell lung (NSCLC), and endometrial cancers.
Exelixis also had a previous licensing deal with Swedish Orphan Biovitrum (Sobi), which will be canceled now it has sealed a deal with Ipsen.
Cabozantinib really has a lot of potential in the Oncology field, and Ipsen seems to have bagged itself a bargain.
Feature Image Credit: © Lexxlam (BigStock ID99063215)