The FDA has cleared Genfit to kick off clinical trials to see if its NASH drug, elafibranor, can also help children.
Genfit develops diagnostic and therapeutic tools for metabolic and inflammatory diseases, particularly those affecting the liver. Its lead candidate, elafibranor, targets Non-Alcoholic Stato-Hepatitis (NASH), and the company’s plan to begin testing it in children has been supported by the FDA. The regulatory body’s decision was based on elafibranor’s good efficacy, safety, and tolerability results in adult patients at Phase IIb. This news has seen the company’s stock rise by 8% since Monday morning and this trend looks set to continue.
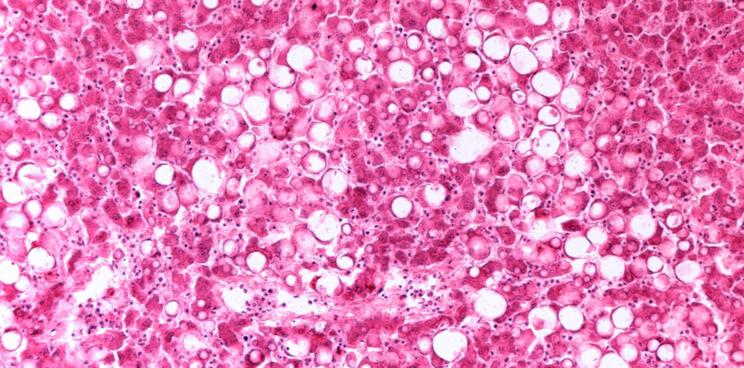
NASH is characterized by the buildup of fat in the liver, which causes inflammation and cell damage, and can even lead to liver failure. Rising rates of obesity and type 2 diabetes are a major public health issue, and they’re contributing to the increased diagnosis of NASH, including in pediatric patients. With no treatments available and the NASH market estimated to be worth up to €37B, it is now a very attractive target for biotechs around the world. With Genfit’s biggest competitor Intercept suffering a major setback, the company leads the field and further strengthened its position by raising €180M in October.
Image – Jose Luis Calvo / shutterstock.com