Epigenomics (Germany and US) has closed a deal to develop and commercialize a new blood-based lung cancer diagnostic in China, one of its highest performing markets.
There are many biotech efforts to find treatments for cancer, but a separate strategy to lower cancer mortality is to improve diagnosis – so that people can be treated when their cancers are still in early stage and manageable.
This is exactly what Epigenomics, one of our favorite Biotechs in Berlin, is doing. Based on their expertise in DNA methylation, one of the most studied forms of epigenetic alterations, Epigenomics is developing ‘liquid biopsies‘ – blood-based tests that can diagnose cancer quickly based on presence of certain biomarkers.

Its first product, Epi proColon, is a test for colorectal cancer that is approved for commercialization in Europe since 2012, while its authorization in the US is still being reviewed after the completion of the ADMIT trial. In China, the test was approved in 2014, after a licensing deal with BioChain, a manufacturer in the Life Sciences field.
China quickly became an important market for Epigenomics (especially as US approval drags on), accounting for more than 50% of its revenues in 2015. So, it’s no wonder that Epigenomics wants to follow the same recipe with another product in development, which detects lung cancer. This means a new deal with BioChain – for an undisclosed value.
BioChain will be responsible for conducting clinical trials in China, which should start already in 2016, and seek approval from the China Food and Drug Admnistration (CFDA). Epigenomics retains the commercialization rights in other territories.
Epigenomics already has a product for lung cancer detection (Epi proLung), but it analyses the bronchial fluid, meaning that patients have to undergo a bronchoscopy. The novel diagnostic (which is the focus of this new deal) will require only a blood analysis.
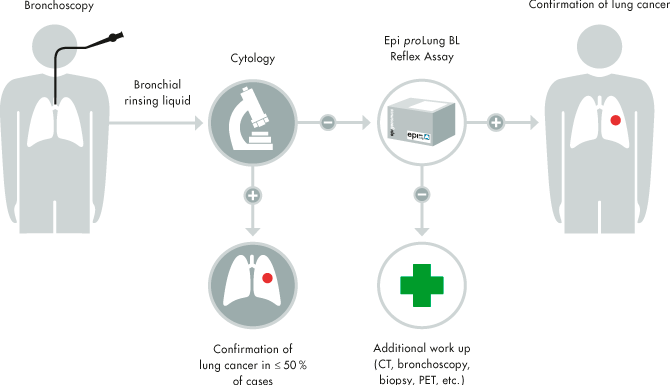
This diagnostic will be based on a range of epigenetic biomarkers, including SHOX2 and PTGER4, two genes involved in the regulation of DNA transcription. Besides the deal with BioChain, Epigenomics has received €2.8M from the Horizon 2020 (the EU’s grant programme) for the development of this blood-based lung cancer test.
Lung cancer is one of the most common and deadly cancers in the world – and its incidence in China is growing rapidly… If the development of this diagnostic test goes well, it could have a big impact on Epigenomics’ finances.





