Islexa is the new baby of Catapult and the University of Aberdeen, which will be developing laboratory grown islets. This could replace insulin production in patients with type 1 diabetes.
![]() One of the coolest Biotechs in London, Catapult has joined the University of Aberdeen to create a new Biotech that could revolutionize treatment of type 1 diabetes (T1D).
One of the coolest Biotechs in London, Catapult has joined the University of Aberdeen to create a new Biotech that could revolutionize treatment of type 1 diabetes (T1D).
Islexa is built on research from University of Aberdeen, which looked into re-programming normal pancreatic tissue into fully functional islets – clusters of cells that produce insulin.
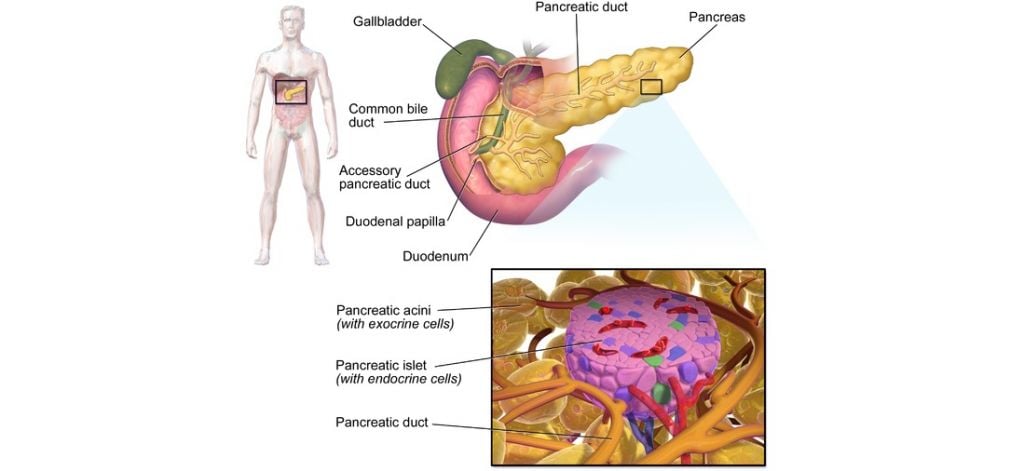
This technology could offer an alternative to patients with T1D. In this type of diabetes, the immune system attacks the cells that produce insulin – one example of harmful inflammation.
Islexa’s laboratory grown islets are meant to bring islet transplants to more diabetes patients. While this procedure has shown efficacy, there is a shortage of suitable donor organs – and the extraction of islets is also complicated.
Islet transplants are particularly important for patients with hypoglycemic unawareness, that have no symptoms when their blood sugar levels drop. This happens to 10% of all T1D patients, and they often have severe episodes – which can result in loss of consciousness and coma.
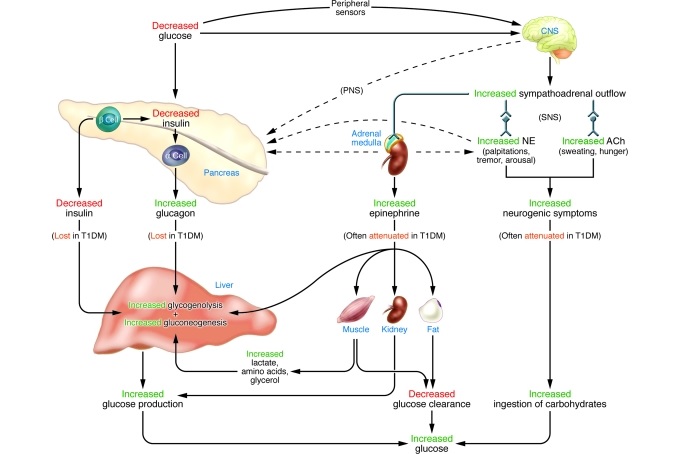
New MedTech is emerging to electronically monitor and manage the disease – such as Cambridge’s bionic pancreas and Cellnovo’s mobile insulin pump. But islet transplants are effective long term, so patients would not need regular insulin injections.
The creation of Islexa follows successful results in pre-clinical studies, which the new company will explore further. The next stage is to take the technology into clinical trials, which are expected in the next few years.
Islexa is maintaining its support from Catapult, which helps cell therapies break into the market, and University of Aberdeen. Other partners include the Scottish Islet Transplant Programme, University of Edinburgh and the Scottish National Blood Transfusion Service (SNBTS).
Islexa’s technology seems promising, and is targeting a big market – more than 300 million people suffer from type 1 diabetes worldwide.
Featured Image Credit: Diabetes (CC 2.0 Dennis Skley / Flickr)
Figure 1 Credit: Blausen (2014) Blausen gallery. Wikiversity Journal of Medicine (doi: 10.15347/wjm/2014.010)
Figure 2 Credit: Cryer (2008) The Barrier of Hypoglycemia in Diabetes. Diabetes (doi: 10.2337/db08-1084)





