Pneumonia, Meningitis and Sepsis: All these potentially fatal infections could one day be vaccinated against with Cambridge’s ImmBio and their Pneumococcal vaccine, which has now started safety & tolerability trials in the UK.
 ImmBio is based at the Babraham Institute, a unit of the UK’s Biotechnology & Biological Sciences Research Council (and in our Listed on our Top 10 EU Bioincubators Ranking) in Cambridge (UK). It was founded based on advances in understanding of the immune system, with two distinct technology platforms, ImmBioVax and ImmunoBodies.
ImmBio is based at the Babraham Institute, a unit of the UK’s Biotechnology & Biological Sciences Research Council (and in our Listed on our Top 10 EU Bioincubators Ranking) in Cambridge (UK). It was founded based on advances in understanding of the immune system, with two distinct technology platforms, ImmBioVax and ImmunoBodies.
Pneumococcal pathogens can be commonly carried in the nasal passages of the general population, and when exposed to vulnerable individuals such as young children and elderly can easily lead to active infection. If untreated, such infection can lead to complications with potentially serious outcomes, including meningitis, sepsis, pneumonia, and death in 14% of cases.
ImmBioVax, mimics normal immune responses to a pathogen through use of multi-protein complexes to stimulate an alert response immune system, and is the platform from which Immbio have developed the PnuBioVax candidate.
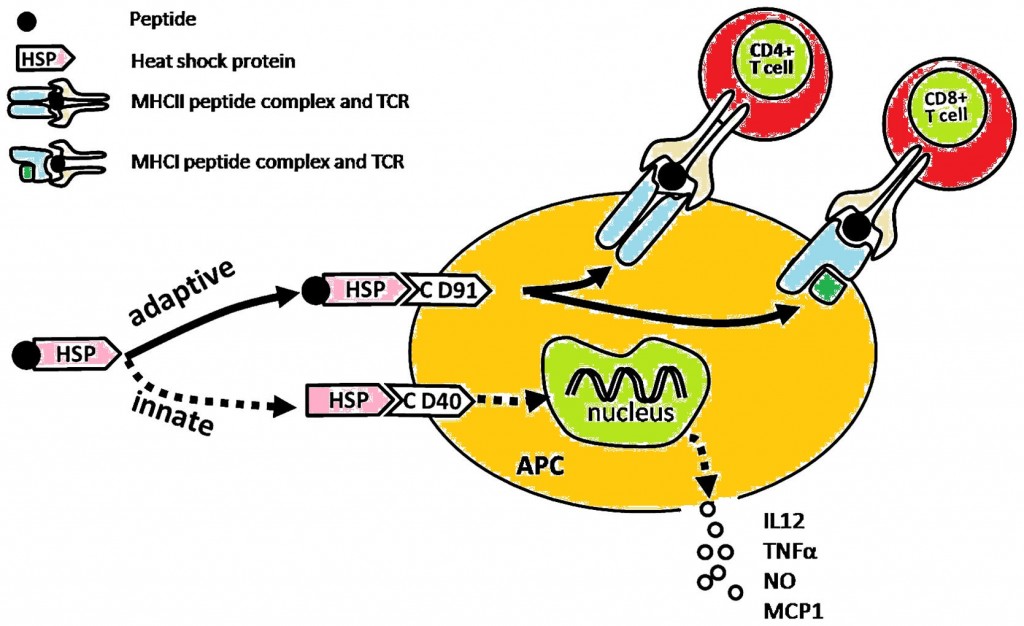
Immbio’s PnuBioVax vaccine contains heat shock proteins (HSPs – also known as chaperones) along with antigens to the Streptococcus Pneumoniae pathogen which primes the immune system against infection from multiple strains – i.e. essentially a universal vaccine for such infections.
The study will look at the safety and tolerability of PnuBioVax for three different dosages at Simbec Research’s site in Glamorgan, Wales (UK), which will enroll 36 volunteers and expects results in Q2 2016. Secondary endpoint measurement focii include the impact on carriage of the pneumococcal pathogen, generation of key antibodies in response to the antigens, and PnuBioVax’s ability to neutralise pneumolysin (a toxin produced by S. pneumoniae).
Other European biotechs looking to fight infections like Pneumonia include Nabriva Therapeutics in Vienna (Austria) which are trialling an entirely new group of antibiotics from funghi (pleuromutilins) to Phase III. There are also various medtech detection assays like the Unyvero System from Curetis (Germany), which is working on earlier diagnosis time of Pneumonia (and therefore quicker response time to treating such hospital acquired infections).
Immbio has partnered its ImmBioVax tuberculosis programme, T-BioVax, with a Chinese vaccine organisation, the China National Biotech Group. The company’s lead internal ImmBioVax program, MenBioVax, targets invasive meningococcal disease, and is co-funded by the UK Biomedical Catalyst.
So the theory sounds solid, and pre-clinical results are generally quite optomistic, but I suppose this UK biotech still has a lot to prove in its first human trials…Here’s to hoping PnuBioVax pulls through.
Figure 1. Zhang & Zheng (2013), Oncology Letters,6(6) 1543-1549 doi: 10.3892/ol.2013.1616