Qiagen is partnering up with Therawis to develop a diagnostic test to predict how effective an aggressive chemotherapy will be in patients with high risk breast cancer.
 A giant in molecular diagnostics, Qiagen is now working in a new diagnostic test that could help guide the treatment of breast cancer.
A giant in molecular diagnostics, Qiagen is now working in a new diagnostic test that could help guide the treatment of breast cancer.
Now, Qiagen has partnered with Therawis Diagnostics, a Munich-based company focused in oncology and predictive tests. Its most advanced programme is to predict how effective certain chemotherapeutic drugs will be against breast cancer.
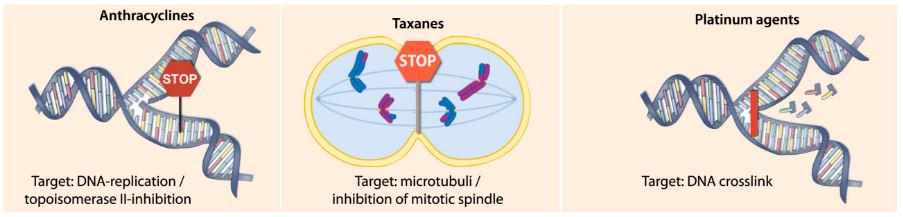
The main focus is anthracycline, a drug currently used to treat patients with high-risk breast cancer. This therapy has serious side effects (such as congestive heart failure or leukemia), and has a relatively low response rate in patients – 50% or less.
A particular type of cancer that has a poor prognosis is triple-negative breast cancer (TNBC). Only about 35% of TNBC patients respond to the chemotherapy, so there’s significant value in a reliable predictive test to guide treatment decisions.
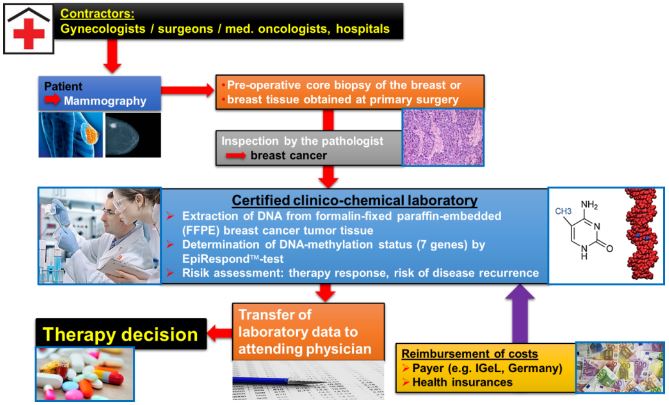
Therawis has achieved initial clinical validation for an assay with PITX2, a transcription factor usually involved in early development – but that could also have a role in helping cells respond to oxidative stress.
This test has demonstrated unprecedented prediction power in triple-negative and other high-risk breast cancers.
With the help of Qiagen’s influence in laboratory equipment, the PITX2 assay can become widely available and part of the standard medical routine in diagnosis – reaching a lot more patients.
Qiagen has been quite active in the field of personalised cancer treatments, with different companion tests for lung cancer (in collaboration agreements with AstraZeneca and US based Mirati) already in its pipeline.





