TolerogenixX has proved that it can get rid of immunosuppressants in organ transplants in Phase I and secured seed funding from High-Tech Gründerfonds.
TolerogenixX is a startup from the Heidelberg University Hospital that develops personalized immunosuppression therapies. Its cell therapy technology has just passed Phase I, where it showed an “impressive efficacy” in preventing the rejection of kidney transplants without the need for immunosuppressive drugs.
The promising results seem to have convinced the German life sciences investor High-Tech Gründerfonds (HTGF), from which TolerogenixX has secured seed funding. HTGF is the first investor to jump in after pre-seed financing from the German Government. The funds, of an undisclosed amount, will help the startup make the preparations for a Phase II trial, planned for spring 2018.
TolerogenixX’s technology provides individualized immunosuppression, tailored specifically to the donor tissue. To do so, peripheral blood mononuclear cells (PBMCs) are harvested from the donor and treated with mitomycin C and then infused into the patient prior to the transplant.
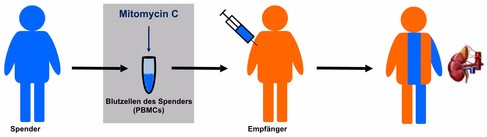
Researchers at the University of Heidelberg discovered that mitomycin C induces a change of behavior in dendritic cells, leading them to suppress T-cell responses. Immunological tests conducted during the trial revealed that the recipients had developed tolerance towards the donor.
“The TolerogenixX technique represents a milestone in the field of individualized immunosuppression,” said Philipp Rittershaus, Investment Manager at HTGF. Indeed, the therapy would allow transplantation without the need for immunosuppressants, which carry many severe side effects and leave patients completely unprotected against infections.
If everything goes well, TolerogenixX expects to complete Phase III and file for approval in 2022. In addition, it will start a second program next year to treat autoimmune diseases such as multiple sclerosis and systemic lupus erythematosus. The methodology would be very similar, just using autologous cells instead for the mitomycin C treatment instead of donor cells.
“This is a real quantum leap in the treatment of transplant patients,” said in a statement Matthias Schaier, CEO of TolerogenixX. “In the future, it will no longer be necessary to take various medications with numerous side effects.”
Images via crystal light / Shutterstock; TolerogenixX