Ariad has presented updated results of an ongoing phase I/II trial for lung cancer, scoring 100% in one-year overall survival.
 Ariad Pharmaceuticals is developing small molecules to treat difficult cancers, using computational and structural biology research. It is based in Cambridge (US) and Lausanne (Switzerland).
Ariad Pharmaceuticals is developing small molecules to treat difficult cancers, using computational and structural biology research. It is based in Cambridge (US) and Lausanne (Switzerland).
One of the hard-to-treat cancers that Ariad is targeting is a type of non-small cell lung cancer (NSCLC), which is characterized by abnormal expression of the proto-oncogene ALK.
This gene codes a protein (anaplastic lymphoma kinase) which is not normally expressed in healthy adult tissues – making it a promising target in Cancer.
Brigatinib is a small-molecule drug taking advantage of this target, and the strategy was recognized as a Breakthrough Therapy by the FDA. Though applicable to a range of cancers with ALK activity (ALK+), the most advanced clinical trials are in NSCLC.
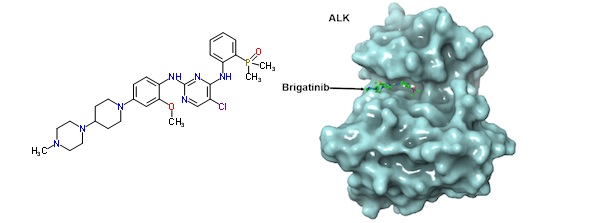
Now, Ariad is presenting its updated results for brigatinib’s phase I/II trial which ran across the US and in 1 centre in Spain.
The highlights are a 100% (8 out of 8) one-year overall survival rate in patients that hadn’t been treat with another type of tyrosine-kinase inhibitor (TKI) – crizotinib (already commercialised by Pfizer).
This seems a big deal, but for the 71-patient group that had previously been treated with crizotinib before (and developed a tolerance) and then Brigatinib this time, the one-year overall survival was 81%. This seems very good.
The candidate has also showed promise with intracranial central nervous system metastases.
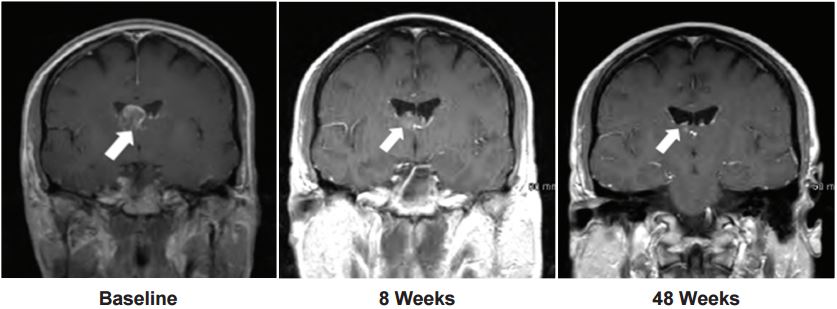
Of the patients with measurable metastases, 53% (8 out 15) showed a significant reduction in size when treated with Brigatinib. And 33% (11 out of 33) of those with non-measurable metastases had complete disappearance of lesions altogether!
Ariad has an ongoing phase II trial exclusively for patients whose disease has progressed on therapy with crizotinib, and is planning a phase III trial this month. Ariad’s CSO, Timothy Clackson, said they expect to submit a new drug application (NDA) later this year.
Along with Mirati’s phase II drug, targeting specific genetic profiles in this deadly type of lung cancer is getting good results.





