AFFiRiS has received €10M from previous investors and one additional backer, adding up to €130M in total funding (half from grants and licensing incomes). This money fuels a pipeline in cardiometabolic and neurodegenerative diseases as more candidates break into clinical development.
![]() Based at the Campus Vienna Biocenter (Austria), AFFiRiS is developing immunotherapies to prevent chronic diseases. Some of its targets are high cholesterol, diabetes, Alzheimer’s and Parkinson’s.
Based at the Campus Vienna Biocenter (Austria), AFFiRiS is developing immunotherapies to prevent chronic diseases. Some of its targets are high cholesterol, diabetes, Alzheimer’s and Parkinson’s.
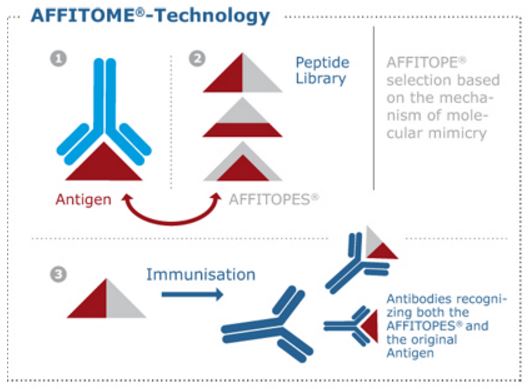
Its vaccines are short synthetic peptides created with Affitome, AFFiRiS’ proprietary platform. These short peptides function as B-cell epitopes, mimicking the part of a protein that antibodies would recognize as a threat. As a result, they can create an immune response against proteins related to diseases (also a popular strategy in allergy, for example).
This new €10M round from AFFiRiS follows another €10M round made in June 2015. One of its existing backers are the Strüngmann brothers (via Santo Holding), who are famous for having sold the generic Pharma Hexal to Novartis for $7.5Bn in 2005. They are one of the big German non-VC investors in biotech with Dietmar Hopp, one of the founders of SAP.
The new fundraising is accompanied by a stronger focus on development, as two candidates in cardiometabolic diseases are now starting Phase I trials.

These new candidates are AT04A and AT06A (for hypercholesterolemia and atherosclerosis), which target PCSK9 enzyme. This enzyme binds to LDL cholesterol (known as ‘bad cholesterol‘) receptors in the liver. With PCSK9 blocked, more receptors are available to remove LDL cholesterol from the blood.
Besides cardiometabolic conditions, AFFiRiS is also contributing to new therapies in neurodegenerative diseases. In particular, their anti-alpha-synuclein vaccine has received a lot of attention in the field Parkinson’s disease.
Feature Image Credit: Plasma cells segregating antibodies © animaxx3d (BigStock ID67843756)





