Vivet Therapeutics has raised a huge €37.5M in Series A to help bring a new gene therapy technology for rare liver diseases to the clinic.
Vivet Therapeutics has managed to attract some of the hottest biotech investors in Europe in its Series A. Led by Novartis Venture Fund and Columbus Venture Partners, and also counting with Roche Venture Fund, HealthCap, Kurma Partners and Ysios Capital, the round has injected a massive €37.5M in the early-stage biotech.
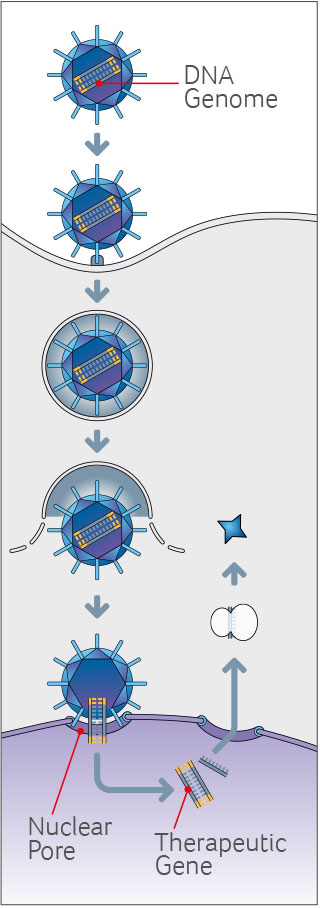 Founded in 2016, Vivet develops gene therapies based on technology in-licensed from the non-profit Fundación para la Investigación Médica Aplicada (FIMA), associated with the University of Navarra, Spain. The technology is based on a next-generation adeno-associated virus (AAV) optimized for increased gene expression in the liver while reducing immunogenic responses.
Founded in 2016, Vivet develops gene therapies based on technology in-licensed from the non-profit Fundación para la Investigación Médica Aplicada (FIMA), associated with the University of Navarra, Spain. The technology is based on a next-generation adeno-associated virus (AAV) optimized for increased gene expression in the liver while reducing immunogenic responses.
Vivet Therapeutic’s lead program, VTX801, targets Wilson Disease, a rare disorder that only affects around 15,000 people in Europe. It is caused by a defective gene encoding the ATP7B protein implicated in copper metabolism in the liver. Patients suffering from Wilson Disease accumulate copper, which causes severe damage to the liver and the brain. Current treatments are not effective in 45% of patients and do not completely eliminate neurological symptoms.
With the new funds, Vivet expects to be able to initiate the first human trials with VTX801 in Wilson disease by the end of 2018. Its pipeline includes additional rare diseases affecting the liver such as progressive familial intrahepatic cholestasis (PFIC) type 2 and type 3, as well as citrullinemia type I.
The young biotech will have to compete with the Swedish Wilson Therapeutics for the few patients across the world suffering from Wilson Disease. Of note, both companies are funded by HealthCap. Although Wilson Therapeutics is ahead with a Phase II trial for its small molecule candidate, Vivet seems to be the only player so far going for gene therapy. Such an approach would eliminate the need for daily dosing that translates into the current poor adherence shown among patients with Wilson Disease.
Images from Kjpargeter /Shutterstock; Vivet Therapeutics